
The XeF 2 Molecular Geometry If we want to learn more about how a molecule’s interior appears, we must research molecular geometry. The molecular geometry of XeF 2 is linear because the bond angle between the two pairs coupled with the centre atom is 180 degrees. The hybridization number is 5, thus the hybridization of BrF 3 is s p 3 d. Compared to the bond pairs, the lone pairs are in an equatorial location.The number of valence electrons in Br is 7 and there are 3 monovalent atoms.This chemical compound is not so commonly discussed yet it holds importance in the chemistry. It appears as a colorless-to-white crystalline solid at r.t.p. XeF 2 is the chemical formula for xenon difluoride. Step 3: Calculation for Hybridisation of BrF 3 Xenon difluoride (XeF2) Lewis dot structure, molecular geometry or shape, electron geometry, bond angle, formal charge, hybridization. Where V is the number of valence electrons, M is the monovalent atom attached to the central atom, C is the positive charge and A represents the negative charge.“Hybridization is defined as the process of intermixing of the orbitals of slightly different energies so as to redistribute their energies, resulting in the formation of a new set of orbitals of equivalent energies and shape.”.JEE Main 2022 Question Paper Live Discussion.Difference Between Selling And Marketing.The molecule is polar due to the asymmetrical arrangement of the fluorine atoms around the central bromine atom. TS Grewal Solutions Class 11 Accountancy According to the VSEPR theory, the electron pair geometry of Brf3 is trigonal bipyramidal, while the molecular geometry is T-shaped. 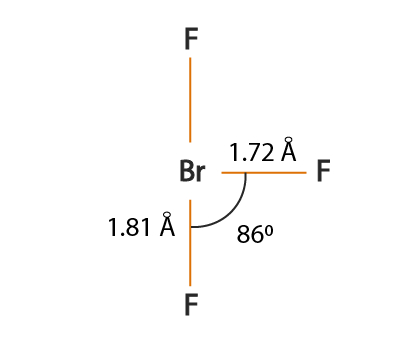
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
